An Accurate and Efficient Look-up Table Equation of State for Two-Phase Compressible Flow Simulations of Carbon Dioxide | Industrial & Engineering Chemistry Research
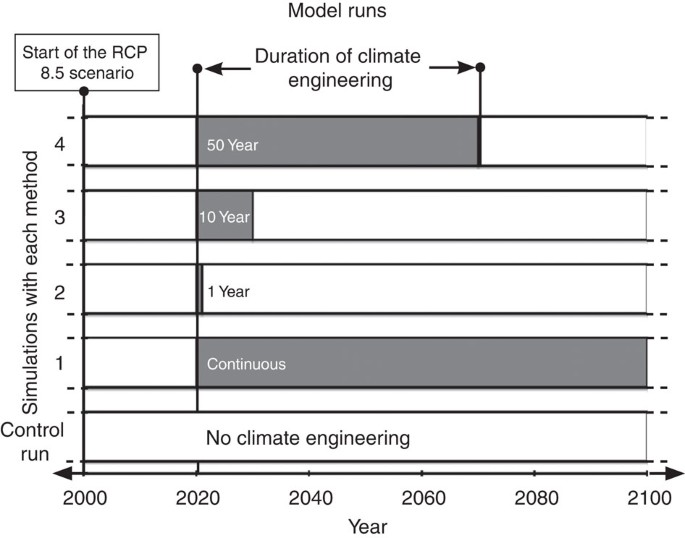
Potential climate engineering effectiveness and side effects during a high carbon dioxide-emission scenario | Nature Communications

ACT Practice Questions.docx - ACT Practice Questions A scientist studying hemoglobin investigated the impact of temperature and carbon dioxide CO2 | Course Hero

A Step toward the Quantification of Noncovalent Interactions in Large Biological Systems: The Independent Gradient Model-Extremely Localized Molecular Orbital Approach | Journal of Chemical Information and Modeling

The maximum work done in expanding 16g oxygen at 300K and occupying a volume of 5dm^3 isothermally until the volume become 25dm^3 is:

Calculate work done when 2 mole of an ideal gas expands isothermally and reversibly at 300 K from 10 atm pressure to 2 atm pressure.

Calculate the amount of carbon dioxide that could be produced when (i) 1 mole of carbon is burnt in air.(ii) 1 mole of carbon is burnt in 16 g of dioxygen.(iii) 2

Methoxy Groups Increase Reactivity of Bifunctional Tetraarylphosphonium Salt Catalysts for Carbon Dioxide Fixation: A Mechanistic Study | The Journal of Organic Chemistry

Calculate the volume occupied by 8.8g of CO2 at 31.1C and 1 bar pressure. R= 0.083 bar dm3 K-1 mol-1 - YouTube

PDF) Oxygen isotope anomaly in tropospheric CO2 and implications for CO2 residence time in the atmosphere and gross primary productivity
How to calculate the work done if three moles of an ideal gas are compressed reversibly and isothermally from a volume of 10 litres to 5 litres - Quora

Metal–CO2 Electrochemistry: From CO2 Recycling to Energy Storage - Wang - 2021 - Advanced Energy Materials - Wiley Online Library